Periodic Table of Elements
*A Picture of the Periodic Table of Elements. √
*Explain what periods are and what they tell you about those elements in each period. √
*Explain what groups or families are and what they tell you about those elements in each period. √
*Explain how elements are classified in the Periodic Table as:
-Valence Electrons (how do you know the # of valence electrons) √
-Metals (Where are metals found?) √
-Nonmetals (Where are nonmetals found?) √
-Metalloids (Where are nonmetals found?) √
-Atomic Number (What does the atomic # tell us about the element and how is it arranged on the periodic table?) √
-Atomic Mass (Where is the atomic mass found and what does it tell us about the element?) √
*Explain how you know if elements are highly reactive or not. √
*List the properties of metals, nonmetals, and metalloids using physical properties such as luster conductivity, and
malleability. √
*Explain what periods are and what they tell you about those elements in each period. √
*Explain what groups or families are and what they tell you about those elements in each period. √
*Explain how elements are classified in the Periodic Table as:
-Valence Electrons (how do you know the # of valence electrons) √
-Metals (Where are metals found?) √
-Nonmetals (Where are nonmetals found?) √
-Metalloids (Where are nonmetals found?) √
-Atomic Number (What does the atomic # tell us about the element and how is it arranged on the periodic table?) √
-Atomic Mass (Where is the atomic mass found and what does it tell us about the element?) √
*Explain how you know if elements are highly reactive or not. √
*List the properties of metals, nonmetals, and metalloids using physical properties such as luster conductivity, and
malleability. √
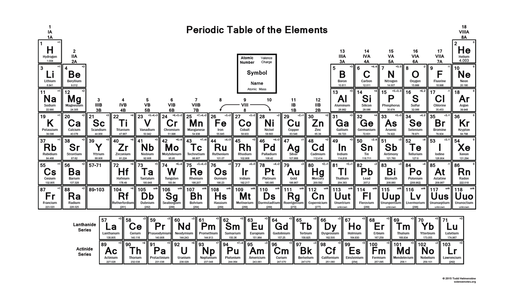
Periods: The rows called "Periods" on the table of elements are horizontal, and show how many orbital rings those elements have.
Groups: The rows called "Groups" on the periodic table are vertical, and show how many valence electrons those elements have.
Elements on the periodic table are organized in many different ways.
The atomic number of an element can tell us two different things. It tells us the # of protons and most times also the # of electrons. The atomic number is found above the elements symbol, and is one of the most important parts of the table.
The atomic mass of an element is close to the atomic number, but not quite. While the atomic number tells us the # of protons and electrons it has, the atomic mass tells us the # of protons and neutrons that are combined. The mass is found below the elements symbol, and is a very important part of the table just like it partner in crime, the atomic number.
Like I said earlier, the groups are vertical and tell us the # of valence electrons it has. The # of valence electrons tells us the # of electrons in the last orbital ring.
Metals, metalloids, and non-metals make up the periodic table with few exceptions. Most of the elements on the table are metals that are found mainly in the center. The metalloids are the elements touching the stair step, and the way I think of them, is that they just can't decide if the want to be a metal or a non-metal, because they can be either one. The non-metals have a much smaller quantity than metals, and are located in the upper right hand corner.
The closer you get to the center of the table, the less reactive you get. For example, Lithium is much more reactive than Nickel or Cobalt.
Groups: The rows called "Groups" on the periodic table are vertical, and show how many valence electrons those elements have.
Elements on the periodic table are organized in many different ways.
The atomic number of an element can tell us two different things. It tells us the # of protons and most times also the # of electrons. The atomic number is found above the elements symbol, and is one of the most important parts of the table.
The atomic mass of an element is close to the atomic number, but not quite. While the atomic number tells us the # of protons and electrons it has, the atomic mass tells us the # of protons and neutrons that are combined. The mass is found below the elements symbol, and is a very important part of the table just like it partner in crime, the atomic number.
Like I said earlier, the groups are vertical and tell us the # of valence electrons it has. The # of valence electrons tells us the # of electrons in the last orbital ring.
Metals, metalloids, and non-metals make up the periodic table with few exceptions. Most of the elements on the table are metals that are found mainly in the center. The metalloids are the elements touching the stair step, and the way I think of them, is that they just can't decide if the want to be a metal or a non-metal, because they can be either one. The non-metals have a much smaller quantity than metals, and are located in the upper right hand corner.
The closer you get to the center of the table, the less reactive you get. For example, Lithium is much more reactive than Nickel or Cobalt.
- Metals are generally shiny, malleable, and hard. Metals are also good conductors of electricity.
- Non-metals do not conduct heat or electricity very well.
- Metalloids share characteristics of both metals and non-metals and are also called semimetals.
